| ANAESTHESIA PEARLS |
- Practice Guidelines for Obstetric Anesthesia
- Ganglion Block Beats Blood Patch for Dural Puncture Headache
| An Updated Report by the American Society of Anesthesiologists Task Force on Obstetric Anesthesia and the Society for Obstetric Anesthesia and Perinatology | |||||
| Summary of Recommendations | |||||
 |
|||||
| 1. PERIANESTHETIC EVALUATION AND PREPARATION | |||||
| History and Physical Examination | |||||
|
• Conduct a focused history and physical examination before providing anesthesia care. * This should include, but is not limited to, a maternal health and anesthetic history, a relevant obstetric history, a baseline blood pressure measurement, and an airway, heart, and lung examination, consistent with the American Society of Anesthesiologists (ASA) “Practice Advisory for Preanesthesia Evaluation.” * When a neuraxial anesthetic is planned or placed, examine the patient’s back. * Recognition of significant anesthetic or obstetric risk factors should encourage consultation between the obstetrician and the anesthesiologist. • A communication system should be in place to encourage the early and ongoing contact between obstetric providers, anesthesiologists, and other members of the multidisciplinary team. | |||||
| Intrapartum Platelet Count | |||||
|
• The anesthesiologist’s decision to order or require a platelet count should be individualized and based on a patient’s history (e.g., preeclampsia with severe features), physical examination, and clinical signs. * A routine platelet count is not necessary in the healthy parturient. | |||||
| Blood Type and Screen | |||||
|
• A routine blood cross-match is not necessary for healthy and uncomplicated parturients for vaginal or operative delivery. • The decision whether to order or require a blood type and screen or cross-match should be based on maternal history, anticipated hemorrhagic complications (e.g., placenta accreta in a patient with placenta previa and previous uterine surgery), and local institutional policies. | |||||
| Perianesthetic Recording of Fetal Heart Rate Patterns | |||||
|
• Fetal heart rate patterns should be monitored by a qualified individual before and after administration of neuraxial analgesia for labor. * Continuous electronic recording of fetal heart rate patterns may not be necessary in every clinical setting and may not be possible during placement of a neuraxial catheter | |||||
| Clear Liquids | |||||
| Aspiration Prevention | |||||
|
• The oral intake of moderate amounts of clear liquids may be allowed for uncomplicated laboring patients. • The uncomplicated patient undergoing elective surgery may have clear liquids up to 2 h before induction of anesthesia. * Examples of clear liquids include, but are not limited to, water, fruit juices without pulp, carbonated beverages, clear tea, black coffee, and sports drinks. * The volume of liquid ingested is less important than the presence of particulate matter in the liquid ingested. • Laboring patients with additional risk factors for aspiration (e.g., morbid obesity, diabetes mellitus, and difficult airway) or patients at increased risk for operative delivery (e.g., nonreassuring fetal heart rate pattern) may have further restrictions of oral intake, determined on a case-by-case basis. | |||||
| Solids | |||||
|
• Solid foods should be avoided in laboring patients. • The patient undergoing elective surgery (e.g., scheduled cesarean delivery or postpartum tubal ligation) should undergo a fasting period for solids of 6 to 8 h depending on the type of food ingested (e.g., fat content). | |||||
| Antacids, H2-receptor Antagonists, and Metoclopramide | |||||
|
• Before surgical procedures (e.g., cesarean delivery and postpartum tubal ligation), consider the timely administration of nonparticulate antacids, H2-receptor antagonists, and/or metoclopramide for aspiration prophylaxis.
| |||||
| 2. ANESTHETIC CARE FOR LABOR AND DELIVERY | |||||
| Timing of Neuraxial Analgesia and Outcome of Labor | |||||
|
• Provide patients in early labor (i.e., less than 5 cm dilation) the option of neuraxial analgesia when this service is available. • Offer neuraxial analgesia on an individualized basis regardless of cervical dilation. * Reassure patients that the use of neuraxial analgesia does not increase the incidence of cesarean delivery. | |||||
| Neuraxial Analgesia and Trial of Labor after Prior Cesarean Delivery | |||||
|
• Offer neuraxial techniques to patients attempting vaginal birth after previous cesarean delivery. • For these patients, consider early placement of a neuraxial catheter that can be used later for labor analgesia or for anesthesia in the event of operative delivery. | |||||
| Analgesia/Anesthetic Techniques | |||||
| Early Insertion of a Neuraxial (i.e., Spinal or Epidural) Catheter for Complicated Parturients. | |||||
|
• Consider early insertion of a neuraxial catheter for obstetric (e.g., twin gestation or preeclampsia) or anesthetic indications (e.g., anticipated difficult airway or obesity) to reduce the need for general anesthesia if an emergent procedure becomes necessary. * In these cases, the insertion of a neuraxial catheter may precede the onset of labor or a patient’s request for labor analgesia. | |||||
| Early Insertion of a Neuraxial (i.e., Spinal or Epidural) Catheter for Complicated Parturients. | |||||
|
• Consider early insertion of a neuraxial catheter for obstetric (e.g., twin gestation or preeclampsia) or anesthetic indications (e.g., anticipated difficult airway or obesity) to reduce the need for general anesthesia if an emergent procedure becomes necessary. * In these cases, the insertion of a neuraxial catheter may precede the onset of labor or a patient’s request for labor analgesia. | |||||
| Continuous Infusion Epidural Analgesia. | |||||
|
• Continuous epidural infusion may be used for effective analgesia for labor and delivery. • When a continuous epidural infusion of local anesthetic is selected, an opioid may be added to reduce the concentration of local anesthetic, improve the quality of analgesia, and minimize motor block. | |||||
| Analgesic Concentrations. | |||||
|
• Use dilute concentrations of local anesthetics with opioids to produce as little motor block as possible Single-injection Spinal Opioids with or without Local Anesthetics. • Single-injection spinal opioids with or without local anesthetics may be used to provide effective, although timelimited, analgesia for labor when spontaneous vaginal delivery is anticipated. • If labor duration is anticipated to be longer than the analgesic effects of the spinal drugs chosen, or if there is a reasonable possibility of operative delivery, then consider a catheter technique instead of a single-injection technique. • A local anesthetic may be added to a spinal opioid to increase duration and improve quality of analgesia. | |||||
| Pencil-point Spinal Needles. | |||||
|
• Use pencil-point spinal needles instead of cutting-bevel spinal needles to minimize the risk of postdural puncture headache. | |||||
| Combined Spinal–Epidural Analgesia. | |||||
|
• If labor duration is anticipated to be longer than the analgesic effects of the spinal drugs chosen, or if there is a reasonable possibility of operative delivery, then consider a catheter technique instead of a single-injection technique. • Combined spinal–epidural techniques may be used to provide effective and rapid onset of analgesia for labor. | |||||
| Patient-controlled Epidural Analgesia. | |||||
|
• Patient-controlled epidural analgesia (PCEA) may be used to provide an effective and flexible approach for the maintenance of labor analgesia. • The use of PCEA may be preferable to fixed-rate continuous infusion epidural analgesia for administering reduced dosages of local anesthetics. • PCEA may be used with or without a background infusion. | |||||
| 3. REMOVAL OF RETAINED PLACENTA | |||||
| Anesthetic Techniques | |||||
|
• In general, there is no preferred anesthetic technique for removal of retained placenta. * If an epidural catheter is in place and the patient is hemodynamically stable, consider providing epidural anesthesia. • Assess hemodynamic status before administering neuraxial anesthesia. • Consider aspiration prophylaxis. • Titrate sedation/analgesia carefully due to the potential risks of respiratory depression and pulmonary aspiration during the immediate postpartum period. • In cases involving major maternal hemorrhage with hemodynamic instability, general anesthesia with an endotracheal tube may be considered in preference to neuraxial anesthesia. | |||||
| Nitroglycerin for Uterine Relaxation | |||||
|
• Nitroglycerin may be used as an alternative to terbutaline sulfate or general endotracheal anesthesia with halogenated agents for uterine relaxation during removal of retained placental tissue. * Initiating treatment with incremental doses of IV or sublingual (i.e., tablet or metered dose spray) nitroglycerin may be done to sufficiently relax the uterus. | |||||
| 4. ANESTHETIC CARE FOR CESAREAN DELIVERY | |||||
| Equipment, Facilities, and Support Personnel | |||||
|
• Equipment, facilities, and support personnel available in the labor and delivery operating suite should be comparable to those available in the main operating suite. • Resources for the treatment of potential complications (e.g., failed intubation, inadequate analgesia/anesthesia, hypotension, respiratory depression, local anesthetic systemic toxicity, pruritus, and vomiting) should also be available in the labor and delivery operating suite. • Appropriate equipment and personnel should be available to care for obstetric patients recovering from neuraxial or general anesthesia. | |||||
| General, Epidural, Spinal, or Combined Spinal–Epidural Anesthesia | |||||
|
• The decision to use a particular anesthetic technique for cesarean delivery should be individualized, based on anesthetic, obstetric, or fetal risk factors (e.g., elective vs. emergency), the preferences of the patient, and the judgment of the anesthesiologist. * Uterine displacement (usually left displacement) should be maintained until delivery regardless of the anesthetic technique used. • Consider selecting neuraxial techniques in preference to general anesthesia for most cesarean deliveries. • If spinal anesthesia is chosen, use pencil-point spinal needles instead of cutting-bevel spinal needles. • For urgent cesarean delivery, an indwelling epidural catheter may be used as an alternative to initiation of spinal or general anesthesia. • General anesthesia may be the most appropriate choice in some circumstances (e.g., profound fetal bradycardia, ruptured uterus, severe hemorrhage, severe placental abruption, umbilical cord prolapse, and preterm footling breech). | |||||
| IV Fluid Preloading or Coloading | |||||
|
• IV fluid preloading or coloading may be used to reduce the frequency of maternal hypotension after spinal anesthesia for cesarean delivery. • Do not delay the initiation of spinal anesthesia in order to administer a fixed volume of IV fluid. | |||||
| Ephedrine or Phenylephrine | |||||
|
• Either IV ephedrine or phenylephrine may be used for treating hypotension during neuraxial anesthesia. • In the absence of maternal bradycardia, consider selecting phenylephrine because of improved fetal acid–base status in uncomplicated pregnancies. | |||||
| Neuraxial Opioids for Postoperative Analgesia | |||||
|
• For postoperative analgesia after neuraxial anesthesia for cesarean delivery, consider selecting neuraxial opioids rather than intermittent injections of parenteral opioids. | |||||
| 5. POSTPARTUM TUBAL LIGATION | |||||
|
• Before a postpartum tubal ligation, the patient should have no oral intake of solid foods within 6 to 8 h of the surgery, depending on the type of food ingested (e.g., fat content). • Consider aspiration prophylaxis. • Both the timing of the procedure and the decision to use a particular anesthetic technique (i.e., neuraxial vs. general) should be individualized, based on anesthetic and obstetric risk factors (e.g., blood loss) and patient preferences. • Consider selecting neuraxial techniques in preference to general anesthesia for most postpartum tubal ligations. * Be aware that gastric emptying will be delayed in patients who have received opioids during labor. * Be aware that an epidural catheter placed for labor may be more likely to fail with longer post delivery time intervals. * If a postpartum tubal ligation is to be performed before the patient is discharged from the hospital, do not attempt the procedure at a time when it might compromise other aspects of patient care on the labor and delivery unit. | |||||
| 6. MANAGEMENT OF OBSTETRIC AND ANESTHETIC EMERGENCIES | |||||
| Resources for Management of Hemorrhagic Emergencies | |||||
|
• Institutions providing obstetric care should have resources available to manage hemorrhagic emergencies. ºº In an emergency, type-specific or O-negative blood is acceptable. ºº In cases of intractable hemorrhage, when banked blood is not available or the patient refuses banked blood, consider intraoperative cell salvage if available.Equipment for Management of Airway Emergencies • Labor and delivery units should have personnel and equipment readily available to manage airway emergencies consistent with the ASA Practice Guidelines for Management of the Difficult Airway, to include a pulse oximeter and carbon dioxide detector. * Basic airway management equipment should be immediately available during the provision of neuraxial analgesia . * Portable equipment for difficult airway management should be readily available in the operative area of labor and delivery units (table 3). * A preformulated strategy for intubation of the difficult airway should be in place. * When tracheal intubation has failed, consider ventilation with mask and cricoid pressure or with a supraglottic airway device (e.g., laryngeal mask airway, intubating laryngeal mask airway, and laryngeal tube) for maintaining an airway and ventilating the lungs. * If it is not possible to ventilate or awaken the patient, a surgical airway should be performed. | |||||
| 7. CARDIOPULMONARY RESUSCITATION | |||||
|
• Basic and advanced life-support equipment should be immediately available in the operative area of labor and delivery units. • If cardiac arrest occurs, initiate standard resuscitative measures. * Uterine displacement (usually left displacement) should be maintained. * If maternal circulation is not restored within 4 min, cesarean delivery should be performed by the obstetrics team | |||||
| Source: | |||||
|
Practice Guidelines for Obstetric Anesthesia: An Updated Report by the American Society of Anesthesiologists Task Force on Obstetric Anesthesia and the Society for Obstetric Anesthesia and Perinatology. Anesthesiology 2 2016, Vol.124, 270-300. http://anesthesiology.pubs.asahq.org/article.aspx?articleid=2471779 | |||||
| Newer Concepts In Hemodynamic of Neuraxial Anesthesia: | |||||
| Virendra K. Arya MD, FRCPC Winnipeg, Manitoba, Canada | |||||
| Introduction: | |||||
|
Neuraxial anesthesia (NA) has a long history of success. Anesthesiologists master this technique early during their training, with achievement of technical competence (> 90% success rate) after as few as 40–70 supervised attempts. The ease of performance and long history of NA may therefore give the impression of simplicity. However, much has been learned about the complexity of the anatomy, physiology, pharmacology and applications of NA in various clinical situations. | |||||
 |
|||||
| THEORIES OF HEMODYNAMIC CHANGES ASSOCIATED WITH NEURAXIAL ANESTHESIA | |||||
|
In large surveillance studies, the reported incidence of hypotension and bradycardia after NA is 33% in the obstetrical and 13% in the non-obstetric population. There were a number of early theories to explain this phenomenon, much of which has subsequently been refuted. | |||||
|
These included:
1. The direct circulatory effects of local anesthetics. However, the vascular absorption of local anesthetic from cerebrospinal fluid or epidural space does not result in blood levels sufficient to cause any direct circulatory effects; 2. Relative adrenal insufficiency as a consequence of adrenal sympathectomy. However, the acute circulatory changes after NA have been shown to be relatively unrelated to changes in adrenal hormone levels; 3. Skeletal muscle paralysis was postulated to cause pooling of blood, but the reduction in skeletal muscle tone does not fully explain the observed changes; 4. Ascending medullary vasomotor block with concurrent respiratory insufficiency. However, it is not possible because the local anesthetic concentration in the cerebrospinal fluid is insufficient to cause direct medullary, vasomotor and respiratory depression. Respiratory insufficiency is attributable to medullary ischemia resulting from the associated hypotension and diminished brain stem perfusion. Subsequent to these initial theories, it was postulated that the primary cause of NA-induced hypotension was the preganglionic sympathetic nerve blockade. Systemic vascular resistance (SVR) is reduced by 5-20%; Stroke volume (SV) is reduced by 5-25%; Heart rate (HR) is reduced by 5-25% ; Cardiac output (CO) is reduced by 10-30%; and Arterial blood pressure (BP) is reduced by 15-30%. It was proposed that the extent of sympathetic blockade influences the magnitude of change in SVR. The sympathetic outflow from spinal cord is widespread in the thoracolumbar origin (T1 to L5). Hence, the circulatory effects of NA are variable. NA limited to sacral and lumbar dermatomes will not be expected to cause any circulatory alteration, whereas blocks up to the mid-thoracic levels will cause partial sympathetic denervation. High NA may cause complete sympathetic blockade, including sympathetic outflow to the heart (typically arising from T1-T4). We know that BP = CO Х SVR. CO can be affected by changes in 4 variables: preload, afterload, contractility and HR. Sympathetic blockade limited to the lower or mid-thoracic region causes vasodilation in lower extremities, with compensatory vasoconstriction in the upper extremities that is initiated by carotid and aortic arch baroreceptors responding to the fall in BP. Thus, the calculated SVR is unchanged. As the sympathetic blockade progresses to include the entire thoracolumbar outflow, it becomes impossible for compensatory vasoconstriction to occur. Therefore, a low calculated SVR would be an invariable finding after a spinal blockade where the sensory level extends above the T4 level. As previously mentioned, earlier studies reported a decrease in CO and SV after NA. Since there was no evidence that cardiac contractility was depressed by NA and the reduction in CO was disproportionate to decrease in HR, the only remaining explanation for the decrease in CO was a reduction in venous return (VR). Unlike the arterial system, the venous system retains little vascular tone when its sympathetic nerve supply is interrupted. Consequently, it was proposed that this lower pressure system depends on gravitational forces to return blood to the heart and that VR and cardiac preload are both sensitive to mechanical and positional influences during NA. However, this explanation for NA-induced hypotension was unable to explain the observations in some early studies showing an increase in CO despite a decrease in cardiac filling pressures and hypotension after NA. The preload reduction theory proved not to be the answer.. | |||||
| ALTERNATE MODEL | |||||
| As a result, an alternate model that describes different vascular beds within the body, each with its unique vascular resistance, was proposed. According to this model, some vascular beds have long time constants in that blood takes a longer time to traverse the bed and return to the heart e.g. splanchnic circulation. Other vascular beds have fast time constants, with short blood transit times e.g. heart, brain, and muscle. Changes in arterial tone alone may cause a redistribution of blood from slow to fast beds, thus increasing VR and CO. As a result of this redistribution, the observed increase in CO after NA would be explained. This redistribution of blood away from the slow splanchnic circulation to faster beds has been shown after T5 spinal anesthesia in healthy young volnteers. Radionuclide studies during epidural anesthesia have also shown that there is a reduction in the blood volume of thorax, abdomen and arms, and an increase in the blood volume in the denervated legs. Clearly, this model of the redistribution of blood volume between the different vascular beds precludes the simplistic explanation of venodilation and preload reduction as the main cause of hemodynamic side-effects following NA. However, this model also does not fully explain the observed effects during NA. | |||||
| REDUCTION IN HEART RATE | |||||
|
Apart from hypotension, reduction in HR is second common observation during NA. The simplest explanation for bradycardia during NA could be the loss of sympathetic input to the heart, leaving the heart with unopposed vagal parasympathetic innervation. However, the finding of elevated resting HR in denervated, transplanted hearts indicates that under normal resting conditions, the basal parasympathetic tone exceeds basal sympathetic tone. Thus, sympathetic tone is not generally required to maintain normal resting HR, which is less than the intrinsic automatic firing rate of the sinoatrial node. This fact is further supported by the observation that expected doses of beta-adenoreceptor blockade have little influence on the resting HR in the normally innervated heart, but markedly attenuate exercise-induced tachycardia. These observations suggest that the blockade of cardio-accelerator sympathetic nerves is not the cause of bradycardia during NA unless vagal tone to the heart is also activated. Bradycardia during NA block is very responsive to atropine administration; this indicates the critical influence of increased vagal tone in the genesis of the reduced HR. In light of this, it appears that observed factors causing increased vagal tone, including situations such as young age, anxiety and patients with known vagotonia etc. can have a reflex reduction in HR. These factors are of greater importance than the inhibition of sympathetic cardioaccelerator fibers. In addition to the aforementioned vagal mechanisms, there are important intrinsic cardiac reflexes that are volume sensitive. These include: the local Bazolt Zerish reflex that causes an acute reduction in SA node output in response to decreased LV stretch; the reverse Bainbridge reflex that causes an increase in vagal tone in response to reduced RA stretch; and un-named intrinsic cardiac reflexes which result in bradycardia through intrinsic chronotropic stretch receptors in the heart. The apparent benefit of these regional survival reflexes to the heart is that with decreased volume load heart will need to do less work (decrease HR). However, these HR reducing reflexes in response to acute preload reduction manifest only in absence of an activated sympathetic nervous system by baroreceptor reflex (tachycardia response to hypotension). This bradycardia response can also be seen in conditions of spinal shock post spinal cord injury. Preload reduction theory was used to explain bradycardia associated with NA on the basis of decreased VR triggering these reflexes in absence of increased sympathetic tone. Therefore, based on the “inaccurate” concept of preload reduction as the primary mechanism for hypotension and bradycardia, the management of NA-induced hypotension included preload augmentation with prehydration, head down position of 10-200, left uterine displacement in pregnant patients as primary intervention and the use of vasopressors and atropine if HR is reduced as secondary intervention. Also based on these inaccurate concepts was the teaching to avoid NA in obstructive cardiac lesions (aortic stenosis, mitral stenosis, hypertrophic obstructive cardiomyopathy), complex congenital disorders, severe pulmonary hypertension and volume-depleted states such as pregnancy-induced hypertension. Since the maintenance of preload and afterload is critical in these conditions, the main concern has been that these patients would become hemodynamically unstable due to rapid preload reduction. | |||||
| CONTEMPORARY CONCEPTS OF HEMODYNAMICS ASSOCIATED WITH NEURAXIAL ANESTHESIA: | 1. There is no decline in CO after NA: | ||||
|
Despite earlier studies on the hemodynamic effects of NA conducted in human volunteers reported a 10-30% decrease in CO, CVP and BP some studies at this time also refuted the decrease in CO as a possible mechanism of hypotension after NA. However, these were not considered important; the prevailing opinion on this subject remained that CO was reduced secondary to preload reduction. Bonica and colleagues observed an unexpected increases in CO, HR and SV with high T2-3 blocks; they considered it to be unexplained. The proponents of the preload reduction theory suggested three ways to prevent NA associated hypotension: 1) Infusion of crystalloid or colloid to compensate for the venous blood trapped in the legs. However, initial reports of success in preventing hypotension were not replicated in subsequent studies, despite volumes up to 30ml/kg; 2) Leg compression. It has proven to be quite ineffective, despite the success of the anti-G suite in preventing lower limb pooling and hypotension in aerospace medicine; 3) Lateral uterine displacement was advocated to reduce IVC compression. Although widely used, this procedure is variably applied, and does not reliably prevent hypotension after spinal anaesthesia. It is therefore clear that therapies based primarily on the concept of preload and CO reduction do not reliably prevent hypotension after NA. The studies on hemodynamic changes after spinal anesthesia in severe preeclampsia have shown that these patients are far less likely to develop hypotension than normal pregnant or nonpregnant women. Despite the contracted blood volume in these pre-eclamptic patients, CO is increased after the spinal block. In the last three decades, many studies have showed that CO increases after NA in volume replete subjects, and further volume loading can further increase CO by up to 43% above baseline. Despite this, arterial hypotension will still occur. Studies on high spinal for cardiac surgery have also revealed that CO and preload are increased following NA. In these studies, head-down tilt was utilized to achieve a high block to T1. These observations when considered collectively have led to the focus on the arterial circulation as a source for post NA hypotension in the fluid-replete patients; hypotension after spinal anesthesia primarily reflects a decreased arterial resistance and not a reduction in CO (due to preload reduction). In most cases, VR is maintained and there is a compensatory increase in CO, mediated by an increase in SV and HR. SVR is therefore primarily reduced after NA. | 2. The Guytonian concepts are not applicable in explaining post NA hypotension | ||||
|
It has taken almost 50 years to appreciate that it is afterload reduction that is the cause of NA-associated hypotension. The underlying mechanism for post NA hypotension in the preload reduction theory was that a reduction in CVP would reduce CO, and thus reduces arterial BP. This hypothesis was based on the view that CVP controls CO, as suggested by the experimental studies by Paterson, Starling and Guyton. Guyton and Starling considering RAP/CVP to be an independent variable in their highly artificial experiments is unlikely to be applicable to the intact animal. A recent helpful view is that the volume in the venous system is more relevant than the pressure, and that ‘venous excess’ or ‘stressed volume’ is the important regulating factor on the venous side of the circulation. Venous capacitance and stressed volume are regulated by sympathetic tone in various clinical conditions, such as changing body posture and hemorrhage; these result in reflex increase in sympathetic outflow. After NA, venodilation will be maximal, depending on the location of the veins. If the veins lie below the right atrium, gravity will cause pooling of the blood peripherally, and if the veins are above, there is back-flow of the blood into the heart by gravity. Venous return to the heart, or preload, therefore will depend on patient positioning during NA. The supine posture in humans is unique and different from animals. In humans the supine position of the IVC is gravity dependent, and gravity will help drain blood into the IVC from liver, spleen and mesenteric veins, even if sympathetic squeeze on this splanchnic venous reservoir system is lost due to NA. Only the kidneys and lower limbs lie below the IVC in the human supine position. The lower limbs can trap blood after sympathectomy in supine position because femoral veins pass through inguinal canal, which lie anterior to the IVC and lower limb veins. Trendelenburg positioning of 10-200 will facilitate the VR from lower limbs in humans. Hence, the experiments done in dogs showing a reduction in VR after spinal anesthesia, are not applicable to humans; dogs will lie only in lateral position where the splanchnic circulation will be more dependent in relation to the IVC; pooling of blood will therefore result after the sympathetic block. In pregnant woman lying supine, the splanchnic component of this capacitance will drain directly into the IVC via the hepatic veins; these are not compressed by the uterus and will therefore not affect VR. However, in the head-up position during NA, there will be an augmentation of the arterial dilatation associated hypotension (due to venous pooling leading to VR reduction). In this situation the fall in cardiac filling pressure due to decreased VR may trigger the intracardiac stretch receptors to cause bradycardia in absence of opposing baroreceptor reflex. | 3. Sympathectomy after NA is not complete: | ||||
|
The sympathetic block after NA is rarely complete; some preservation of sympathetic reflexes remains. Endogenous pressor responses seem to be mediated by norepinephrine, which is released by peripheral nerve terminals running along the arterioles that get blocked at spinal level. Arterial tone is consequently reduced after NA. Preganglionic sympathetic outflow to the adrenals is not completely blocked despite T6 to L2 sensory and motor block and postural reflexes remain partially intact. RISK FACTORS FOR NEURAXIAL ANESTHESIA ASSOCIATED HYPOTENSION A high incidence of NA-induced hypotension has been reported in: * Young, * Obese patients (BMI >35); * Patients with hypertension on chronic antihypertensive therapy; * Rapidity of onset of block sensory block above T6 (with in 10 min of procedure); * Chronic alcohol consumption; * Hypovolemia; * Anemia; * ASA 3-4 status; and * Certain types of surgeries (gynecology, general surgery). * High baseline HR, HR variability and high baseline perfusion index (derived from a pulse oximeter) High baseline HR and HR variability indicate a relatively high sympathetic tone or hypovolemia and may help to predict obstetrical patients in whom pre or co-hydration would be beneficial. Baseline perfusion index may reflect basal peripheral arterial tone that is decreased by NA induced sympathectomy. High levels of preoperative anxiety have been shown to have a positive correlation with hypotension after spinal anesthesia in women undergoing caesarean delivery. Serious bradycardia may be seen in patients with autonomic neuropathy, spino-vagal reflex, sitting posture, hypoxia of CNS/heart secondary to hypoventilation or shunting in complex congenital disorders and if initial hypotension is neglected. | NEURAXIAL ANESTHESIA IS SAFE IN CARDIAC DISEASE | ||||
| The traditional cardiac contraindications for NA, especially for spinal anesthesia, have been based on the preload reduction theory and have not been evidence-based. With the new understanding that primary mechanism for hypotension after NA is a reduction in arterial resistance, NA can be given in any cardiac condition, as long as invasive arterial pressure monitoring is used and the physiology is understood. In critical cardiac conditions, vasopressors like phenylephrine infusion can be initiated in low doses of 0.2 mcg/kg/min while spinal anesthesia is being performed and can be titrated to BP. Patient position is maintained in the supine position with 10-200 head down tilt. If the anesthesiologist is particularly concerned about rapid after load reduction, the NA technique may be modified by using a spinal or epidural catheter to allow more controlled onset. However, based on the experience from single shot, high-dose spinal anesthesia in patients with mitral and aortic stenosis undergoing cardiac surgery (with prophylactic vasopressor infusion titrated to invasive BP), sudden severe hemodynamic instability has not been reported. Most anesthesiologists have considered GA to be safer in these patients. However, the induction of general anesthesia similarly produces a degree of central sympathectomy (due to reduced central sympathetic outflow). In addition, positive pressure ventilation and PEEP reduce VR. Induction agents for general anesthesia may have direct myocardial depressant effects; this is not the case with NA. Cardiac anesthesiologists who induce critically ill cardiac patients are well aware of the practice of using prophylactic vasopressor/inotrope infusions and invasive monitoring during the induction of general anesthesia. The same principles should be applied while planning NA in critically ill cardiac patients. Hence, it is largely a myth that general anesthesia is safer than NA in patients with critical cardiac lesions. Both techniques can be safely performed as long as there is invasive BP monitoring and pharmacological dose titration to the hemodynamic endpoints. Currently, high spinal anesthesia has been given safely in cardiac surgery patients for coronary revascularization, severe mitral stenosis for mitral valve replacement and critical aortic stenosis for aortic valve replacement. | MANAGEMENT FOR HEMODYNAMIC CHANGES AFTER NA | ||||
| The current literature suggests that NA-induced hypotension reflects decreased arterial vascular resistance. Therefore, in fluid-replete supine patients, VR is maintained and consequently CO is maintained or increased; there will be an increase in SV and HR. In this situation, a rapid acting alpha-agonist such as phenylephrine is the best option to restore baseline hemodynamics. Preloading the patient with intravenous fluids will further increase in CO, SV and CVP, but the increases are short-lived. The arterial vasodilatation, as is the primary cause of hypotension, remains uncorrected. Moreover, intravenous fluid could cause even further decreases in SVR by hemodilution. It is of course logical to administer fluid before and whilst the block is evolving in relatively volume deplete patients. However, vasopressors should stay as the primary therapeutic option and should be administrated in small intravenous boluses or by infusion to achieve the desired goals; intravenous infusions of these vasopressors is likely more efficient and avoids the sudden peaks and valleys in BP. | CONCLUSION: | ||||
|
1. There is paradigm shift from preload reduction theory to afterload reduction as the primary mechanism of NA induced hypotension. 2. Vasopressors administered as infusions or as titrated bolus are now considered first line therapy in the management of hemodynamic instability secondary to NA. 3. Fluids are only given for volume replacement in hypovolemic patients. 4. There is no myocardial depression with NA; 5. The origin of bradycardia is multifactorial, often has a reflex component, and is usually responsive to atropine. Bradycardia should be treated aggressively before the low CO becomes severe which may in turn delay the circulatory response to therapy. 6. Bradycardia has been identified as the main precursor of cardiac arrest during NA. The early use of epinephrine in severe bradycardia is recommended. 7. The effect of gravity must be considered both for the spread of the block and for its effect on hemodynamics. 8. There seems to be no absolute cardiac contraindications for NA with invasive pressure monitoring, careful positioning and appropriate vasopressor administration. 9. Maintaining adequate spontaneous respiration is crucial for hemodynamic stability after NA. | Source: | ||||
|
Hemodynamics of Neuraxial Anesthesia: Contemporary Concepts. Virendra K. Arya MD, FRCPC Winnipeg, Manitoba, Canada. Refresher Course Lecture. Anaesthesiology 2015. | |||||
| Ganglion Block Beats Blood Patch for Dural Puncture Headache | |||||
| INTRODUCTION: | |||||
| Postdural puncture headache (PDPH), also known as spinal headache, is a common complication associated with neuraxial anesthesia and diagnostic lumbar puncture. Autologous epidural blood patch is considered the definitive treatment for PDPH with efficacy greater than 75%; but it is an invasive procedure that carries the same risks as other epidural procedures, including dural puncture, infection, and neurologic complications. Transnasal sphenopalatine ganglion block (SPGB) has been traditionally used to treat chronic conditions such as migraine, cluster headache, trigeminal neuralgia, and atypical facial pain, and has also been used with good results in a recent case series to treat acute PDPH headache in obstetric patients. | |||||
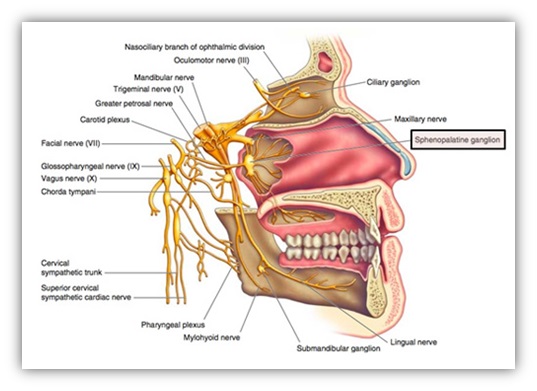
| ANATOMY, NEUROANATOMY AND PHYSIOLOGY OF THE SPHENOPALATINE GANGLION | |||||
 |
|||||
|
The pterygopalatine or sphenopalatine ganglion (PPG, SPG) is one in all four tiny parasympathetic ganglia found within the head. It is the largest parasympathetic ganglion outside the cranium. This square measure (2 in 1) ganglions, placed on both sides of the middle face at the pterygopalatine fossa (PPF). The branches of the pterygopalatine ganglion carry sympathetic, parasympathetic and general sensory fibers. The greater superficial petrosal nerve (GSPN) is a preganglionic parasympathetic nerve from the superior salivatory nucleus in the pons, which via the nervous intermediates of the facial nerves traverse, but not synapse at the geniculate ganglion of the facial nerve. It connects deep petrosal nerve (DPN). Sympathetic fibers from T1 – T3 synapse at the superior cervical ganglion. Sympathetic postganglionic fibers along the internal carotid artery entering the skull as DPN at the proximal part of the canal and form the Vidian nerve, that traverses through the pterygoid canal and reaches the PPF. The ganglion is joined by general sensory fibers from the maxillary branch and these nerves forms the sensory component of the SPG. The trigeminal nerve has no autonomic function. The facial cranial nerve that renders the parasympathetic autonomic secretomotor function utilizes the trigeminal cranial nerve as an anatomical vehicle for its postganglionic parasympathetic fibers. The branches of parasympathetic fibers which innervate the cerebral and meningeal blood vessels activated and released neuropeptides that cause vasodilation and/or activation of trigeminal nociceptor fibers in the meninges. It is perceived as referred pain from the head by the sensory cortex. As a result of the trigeminal-autonomic reflex activation SPG is believed to have a role in headache and cranial autonomic symptoms associated with cluster headache. Cranial parasympathetic outflow contributes to migraine pain by activating or sensitizing (or both) intracranial nociceptors, which induce parasympathetically independent allodynia by sensitizing the central nociceptive neurons in the spinal trigeminal nucleus. SPG has been the site for a variety of clinical interventions for the headaches treatment as a result of the involvement of the SPG within the trigeminal-autonomic pain reflex. Stimulation of the parasympathetic fibers of the sphenopalatine ganglion related to primary headache disorders such as cluster headaches and migraines, trigeminal autonomic cephalgia (TAC) and cerebrovascular regulation (CVR). The sympathetic post ganglion fibers involved in post herpetic neuralgia (PHN), atypical facial pain, and CVR. The trigeminal sensory fiber from maxillary nerve involved in somatosensory pain in headache, trigeminal neuralgia or orofacial pain. Identification of a nerve, which runs between the PPG and the ophthalmic nerve with sensory and parasympathetic fibers may provide an anatomic basis for pain relief in the ophthalmic area after PPG blockage. So three possible mechanisms of sphenopalatine ganglion block (SPGB) are: a) Interrupting the post-ganglionic parasympathetic path that inhibits the pain and cephalic autonomic symptoms, b) The sensory process modulation in the trigeminal nucleus, and c) Block of sympathetic mediated pain due to interruption of postganglionic sympathetic outflow | |||||
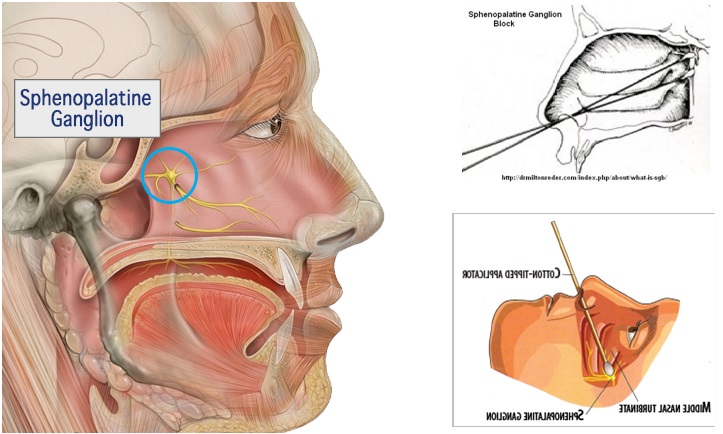
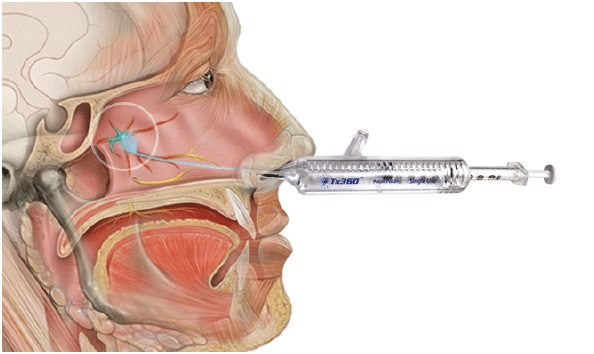
| PROCEDURE: | |||||
 |
|||||
|
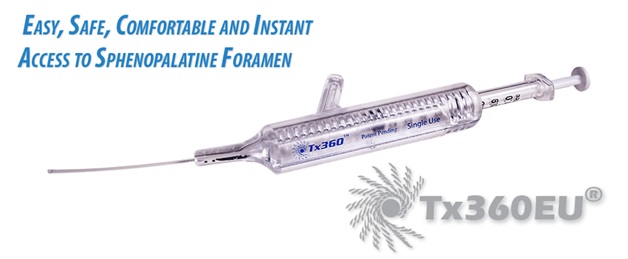
The procedure involves placing two 6-inch, cotton-tipped, hollow plastic applicators bilaterally into each nasal passage, placing the applicator superior to the middle nasal turbinate, and injecting 0.5 to 1.5 mL of 4% lidocaine on each side. Tx360EU® is a single-use, disposable catheter that delivers medication through the nasal passages to a difficult-to-reach ganglion located at the back of the nose easily, safely, efficiently and accurately (only 0.6cc of medication needed). | |||||
 |
|||||
 |
|||||
| EVIDENCE: | |||||
|
Preet Patel, MD et al. Sphenopalatine Ganglion Block (SPGB) Versus Epidural Blood Patch (EBP) for Accidental Postdural Puncture Headache (PDPH) in Obstetric Patients: A Retrospective Observation. Presented at the 2016 AAPM Annual Meeting. This retrospective study evaluates the efficacy of SPGB as an alternative treatment for PDPH. After IRB approval, we reviewed 72 records, over a 17 year period, of parturients without a previous history of primary headaches who had experienced PDPH. Group I parturients (n=33) received SPGB for PDPH. EBP was available for patients upon request. Group II parturients (n=39) had routine EBP for PDPH. Patients were followed up at ½ hr, 1 hr, 24 hr, 48 hr and 1 week (by phone) post-treatment. The two groups had similar baseline characteristics including ASA class, age, height, weight and BMI (p>0.05). At ½ hr. post-treatment, 18/33 patients (54.55%) in Group I had recovered from headache versus 8/39 patients (20.51%) in Group II (p=2.73 x 10-3). At 1 hr. post-treatment, 21/33 patients (63.64%) in Group I had recovered from headache versus 12/39 patients (30.77%) in Group II (p=5.29 x 10-3). At 24 hr., 48 hr. and 1 week post-treatment, no differences were seen. Group II parturients experienced higher complication rates including 9 patient ER visits, 3 complaints of backache radiating to LE, 1 vasovagal reaction and 1 complaint of temporary hearing loss. SPGB is a highly effective treatment for PDPH in obstetric patients. SPGB is a non-invasive treatment with minimal side effects which relieves PDPH faster than EBP and with fewer complications. | |||||
| Tx360EU® Live Instruction Video | |||||